Bring active learning to your lab, classroom, or online course
with our easy-to-use software platforms, interactive content, and hardware
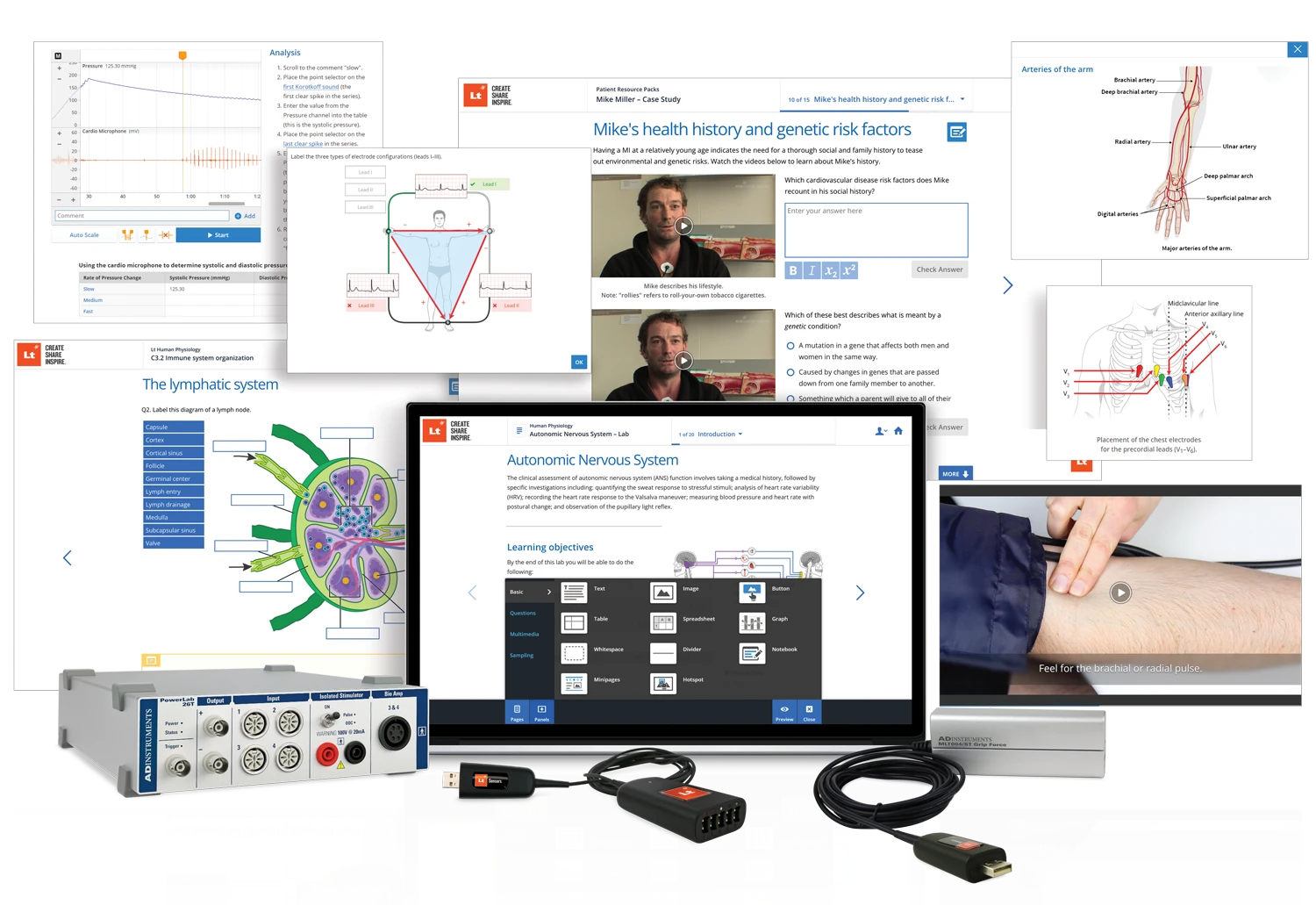
Our customizable education systems provide all the experiments, hardware, software, and lessons you need to teach and create a stimulating learning environment that guides your students to connect theory and practice.
Creating a custom education system is easy
Future-proof your course with our innovative and versatile teaching platform.


"We have seen high student engagement and satisfaction, and widespread adoption over many disciplines and courses."
- Sharon Herkes, Senior Lecturer, Physiology, University of Sydney
Meet our Content Team
Our Education Content Team is a highly skilled and experienced team who are passionate about science and learning! We design and create the lessons and labs within Lt and Lt LabStation, in a variety of subject areas including physiology, anatomy, nursing, medicine. and biology. Our team includes instructional designers, medical illustrators, and a localization expert.
An important aspect of our work is to keep up with the latest learning design strategies and evidence-based teaching practices so that we can apply these strategies and theories to create accessible, interactive lessons. We introduce students to metacognitive skills so that they can take control of their own learning, and further engage them through questions and interactive activities.
Our medical illustrators make sure our lessons are full of beautiful images with a clear and consistent style, while taking accessibility guidelines into account.
“I want to make high quality and engaging material that pushes the boundaries of science education.”
- Tim, Instructional Designer
Alternative software
If you need a lab-based solution, Lt LabStation has all of the benefits of Lt and is ideal for offline life science teaching.
If you want to use research-grade software and hardware in the teaching lab, we offer LabChart for Education.
A cost-effective way for your students to quickly and easily record biosignals directly into Lt via USB.
Lt Sensors connect directly to Lt through the USB connection on your Windows-based laptop or desktop computer. Students use the Lt Sensors to record and analyze their own physiological signals in real time – these products function as ECG/EKG, EEG, EMG, EOG, respiratory rate, and finger pulse sensors. This active engagement helps students to quickly grasp and understand core physiological concepts in Lt’s Human Physiology Labs.
Lt USB Sensors are perfect for educators that require new or additional hardware for human physiology courses. Together with Lt, these sensors are an affordable, easy to use data acquisition solution that give students an authentic lab experience, while also allowing flexibility between face-to-face and remote learning.
High performance data acquisition for your life science classroom experiments
PowerLab is engineered for precise, consistent, reliable data acquisition, giving you the reproducible data you need while meeting the strictest international safety standards. Simple to set up, PowerLab connects directly to Windows and Apple computers via USB port.
The T Series PowerLab is an economically priced high quality DAQ unit with built-in, fully isolated, two-channel bio amplifier, isolated stimulator, separate non-isolated analog inputs and a differential-mode analog output.
"PowerLab simply WORKS. It never crashes or has to be rebooted, and is 100% reliable."
- Dr Geovanni Casotti, Professor of Biology, West Chester University, USA.
Choose from a range of education kits to suit your specific teaching requirements and budget. Created with focus and flexibility in mind, our education kits have been designed to align with the latest Lt and Lt LabStation lessons and content, ensuring that you have everything you need for your teaching labs, and nothing you don’t.
All education kits can be matched with your choice of software, lessons and PowerLab.
















