Reining chaos to save lives
A novel approach, combining maths and medicine to diagnose presymptomatic sepsis earlier, has the potential to save lives.
Dr. Manasi Nandi Ph.D. is a cardiovascular pharmacologist and a Senior Lecturer in Integrative Pharmacology at King’s College London. Manasi’s particular research interest is in sepsis and the cardiovascular dysregulation that occurs in those patients. She is working on a groundbreaking interdisciplinary project with mathematician Professor Philip Aston from the University of Surrey, that aims to detect the onset of disease earlier. In the setting of critical care and sepsis, this could be lifesaving.1

It’s often described as the hidden killer.
This deadly combination of infection and inflammation strikes more than a million Americans every year2, and kills someone every 2 minutes3-4. It kills more people annually in the UK than bowel cancer, breast cancer, and prostate cancer combined5. It’s a leading cause of mortality in children worldwide.6
The killer is sepsis - a broad term for our body’s overwhelming reaction to an infection. The cascade of organ failure caused by severe sepsis and septic shock can be treated and stopped, but only if doctors recognize the symptoms in time - and that’s not easy to do.
Dr Manasi Nandi, a cardiovascular pharmacologist in the Faculty of Life Sciences and Medicine at King’s College London, is working on a joint project with Prof Philip Aston, a mathematician at the University of Surrey, that aims to predict whether a patient is about to crash from sepsis, by analyzing patterns in raw cardiovascular data collected from patients in hospitals.
The team’s project has the potential to help doctors diagnose patients earlier – which could be lifesaving.
Sepsis - A Global Issue
KEY FACTS
The World Health Organization estimates that each year:
1.2 million
children suffer from sepsis
6 million
deaths are caused by sepsis
30 million
people worldwide are affected by sepsis
3 million
newborns suffer from sepsis
1 million
newborn deaths are associated with maternal infection such as maternal sepsis
Hundreds of millions
of patients are affected by health care-associated infections that occur during care delivery
Sepsis: the problem and the challenge.
Sepsis is a life-threatening condition caused by our body’s overwhelming immune response to an infection. Normally, when bacteria or other microbes enter our body, our immune system efficiently destroys the invaders. However, in sepsis, the immune system responds in an extreme way - by going into overdrive and releasing a cascade of inflammatory mediators to combat the infection. However, this process can lead to rapid deterioration of the cardiovascular system.
Manasi explains, “With sepsis, one of the things that particularly happens in the cardiovascular system is that small blood vessels can become leaky as a result of the inflammation. Your heart initially compensates by beating faster to try and maintain the cardiac output, but this can eventually fail. The blood vessels dilate, and the blood pressure drops and together this means that your organs and tissues and cells don’t receive enough oxygen”.
Septic shock is the progression of sepsis to a more life-threatening stage where the cardiovascular system becomes dysfunctional. The goal is to have an earlier warning that a patient is in the initial stages of the sepsis syndrome. A warning would mean the doctors would ‘refill’ the patient by using intravenous fluids or do an earlier blood test to identify the microbe in order to treat with targeted antimicrobial therapies. The key is to try and do this before the syndrome progresses to septic shock.
But sepsis is unpredictable, making it notoriously difficult to diagnose. Patients present with different signs and symptoms at different times. It can be a response to an infection anywhere in the body, from something as simple as an infected cut or insect bite, to an infection caused by pneumonia, influenza, or a urinary tract infection.
Depending on the infection, sepsis can affect any organ, resulting in a range of symptoms. If the brain is affected, this may cause confusion; if the lungs are affected, this may result in breathing difficulties.
When sepsis affects the cardiovascular system, the body’s response can cause rapid deterioration. According to Manasi, “30 to 50% of patients don't survive septic shock, so diagnosing these patients earlier or being able to give clues to a doctor that a patient is about to deteriorate is absolutely critical.”
Infection begins anywhere in the body
Immune system floods bloodstream with inflammatory mediators, damaging blood vessels and causing them to leak
Blood pressure drops dramatically and organs don’t receive enough oxygen, leading to multiple organ failure
Earlier detection of sepsis: The key to survival
It is estimated that as many as 80% of sepsis deaths could be prevented with rapid diagnosis and treatment.7
Because the risk of death from sepsis increases by as much as 8% for every hour that treatment is delayed, the early diagnosis of sepsis is vital for patient survival.8
Earlier detection is ultimately what Manasi’s research project is hoping to achieve. “If we can detect cardiovascular disturbances earlier, then we can bring onboard well-characterized treatments much sooner. But doctors need a signal that tells them that this patient's about to crash, rather than this patient has already crashed.”
The way that Manasi and her team are hoping to do this is by analyzing cardiovascular monitoring data that has been collected from hospital patients, in a new and novel way.
Manasi explains, “we are looking at a variety of cardiovascular signals - like blood pressure or ECG or pulse oximetry data - that have been collected from patients. We're trying to see if there are patterns showing how those waves change in a patient that went on to recover versus a patient that went on to decline and become seriously ill.”
The key is that Manasi and her team don’t only want to use the routine cardiovascular outputs (systolic, diastolic pressure, ECG intervals), but use the entire signal which contains all of the information pertaining to the contours of the waveforms.
We hypothesize, hidden in the signals, is pivotal information that could help to alert doctors that a patient is starting to deteriorate at a stage where it is currently undetectable.
Using Big Data
Modern hospital monitoring devices record a huge amount of high fidelity waveform data about a patient’s body. For instance, a monitoring device that collects blood pressure signals can record as many as 1,000 individual numbers for every single heartbeat.9
Manasi explains, “our research project is very much about using the entirety of the data, the “big data” if you like, to gain a much more detailed picture of cardiovascular health and to get more clues about what's happening to a patient, much much earlier than we can at the moment.”
“The devices that we currently use have a sampling frequency 1,000 Hz, meaning that they capture 1,000 data points per second, which equates to over 86 million data points in one day.”
When all of this data is collected over hours or days it becomes too much to process using conventional methods. Instead, the relevant information is processed and output as averages, minimums and maximums - which we understand as simplified numbers like systolic and diastolic pressure, heart rate, and blood oxygen level.
However, most of the raw data is lost by simplifying it in this way.10
Manasi explains, “we've known for over 150 years that the shape of the waveform contains some useful information and potentially diagnostic information. So you've got a waveform for, say, blood pressure, and in the background you've got all of these numbers that make up that wave. But we don’t really use it all, although we know that it contains important cardiovascular information.”
To glean more information from the raw data in the entire waveform, Manasi partnered with mathematician Professor Philip Aston from the University of Surrey. Their aim was to use mathematics to look at the cardiovascular waveform data differently and find new, creative ways of processing the data as a way to extract more information.
“What this project allows us to do is use every single piece of numerical data that has been captured by the monitoring devices and plot it in a particular way using some clever mathematics. We can handle the big data in a way that is manageable, and it gives us quantitative information from the signal that we previously couldn’t measure. Importantly, we don’t change the raw data, we just visualize it in a different manner.”
Unlocking the diagnostic potential hidden in the entirety of the waveform is what Manasi believes will give health professionals a more sensitive readout of cardiovascular deterioration in patients - and give them the signals to treat patients earlier.
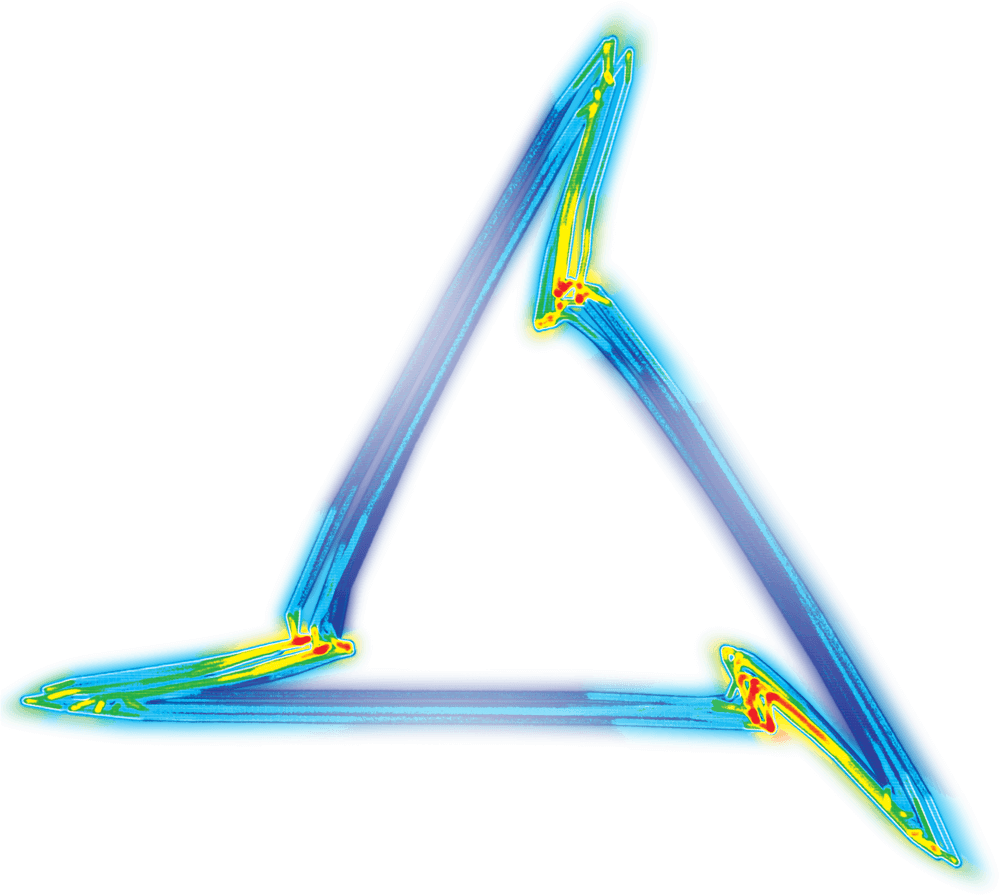
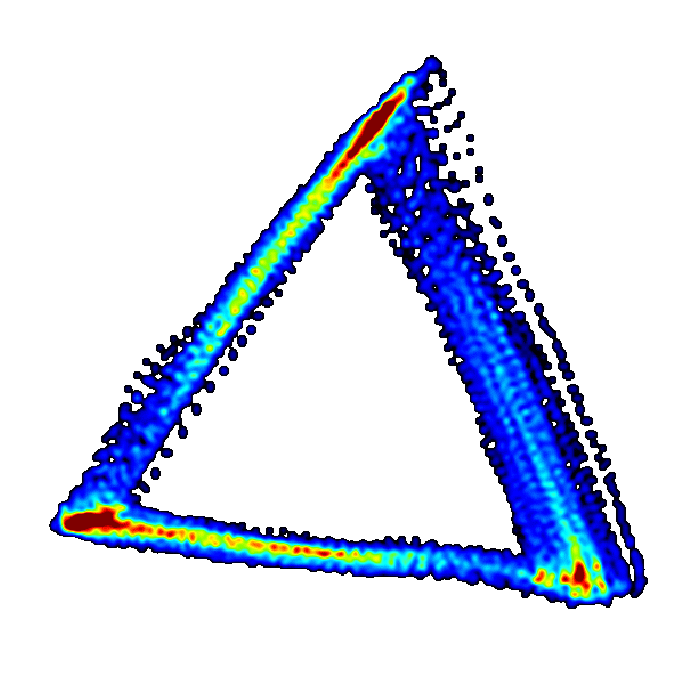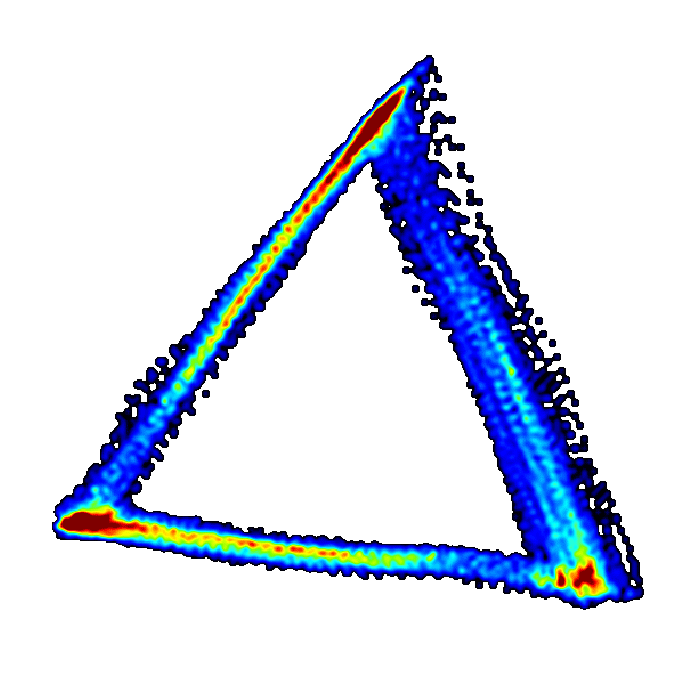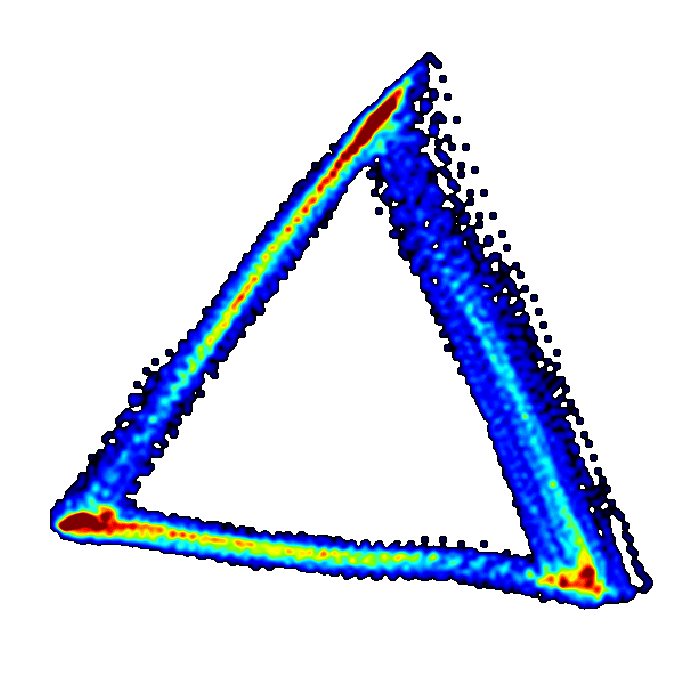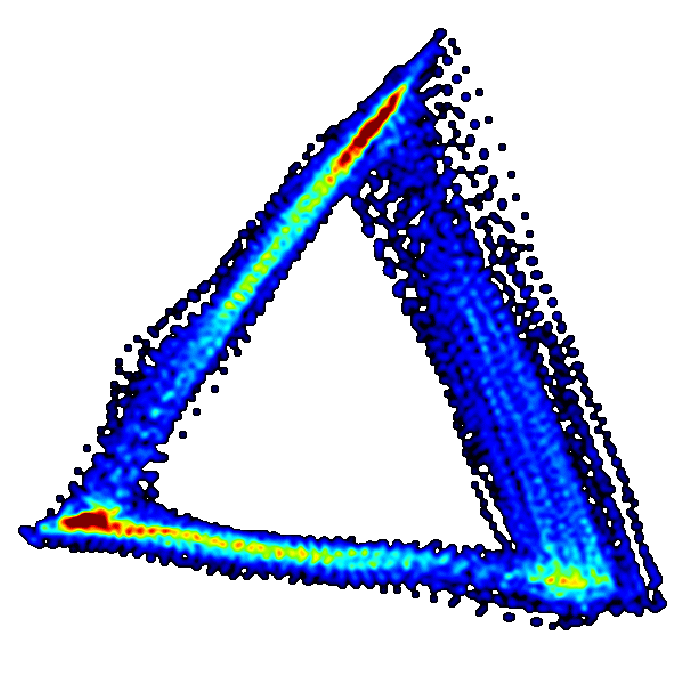
So how does it work? The answer lies in a mathematical attractor called Attractor Reconstruction – but which has been termed a ‘Cardiomorph’.

Creating the Cardiomorph
Manasi’s research partner, University of Surrey mathematician Philip Aston, takes the cardiovascular monitoring data provided by Manasi and applies a mathematical method using Takens’ embedding theorem. The key point is that it does not make any assumption about the data and is able to process noisy signals which are sometimes described as being chaotic.
The method takes the high-fidelity cardiovascular data and re-plots it into a 3-dimensional cube, and this is called plotting in ‘phase space’. This generates a 3D meshwork re-representing the data in its entirety – but at this stage the attractor is a noisy meshwork and it’s difficult to extract any information from it. However, by rotating the cube and looking at the attractor down one corner of the cube, it appears as a triangle. It’s now much easier to extract quantitative information from this triangle – which is called an ‘attractor or ‘cardiomorph’.
The cardiomorph is a new way of visualizing the cardiovascular waveforms. Manasi explains, “there is a one-to-one relationship between the waveform shape and the cardiomorph shape. Tiny changes in the shape of the waveform appear as much bigger changes on the cardiomorph.
“What we have shown so far is that those subtle changes in the shape of the wave give you clues about early stage cardiovascular failure, before obvious changes occur, like your maximum and minimum blood pressure dropping.”
“Plotting the data like this lets us see patterns. It means that we can start to build algorithms that could then be potentially, in a few years’ time, installed as software that gives doctors an alarm signal to tell them that a patient might be deteriorating and may require treatment.
Using data to predict the unpredictable
So, the big question is: can the attractor method be used to diagnose or detect sepsis earlier in patients?
Currently, the team are reliant on using archived data to build their attractor. They are comparing existing data collected from patients who came into hospital – some developed sepsis during their stay, others didn’t. Manasi explains, “we are still very much in the earliest stages of research. We're feeding known data into the system. We use our attractor data combined with the clinical notes - the annotations that the doctor put on the original record - to see if our method could have predicted that a patient was going to deteriorate earlier than the clinical notes suggests.”

By re-examining existing data, they can see if there are any emerging patterns in the attractor. “If we’re successful at finding attractor signatures that correlate with particular patients, then we can start to build algorithms that could be installed into existing software and used in a clinical environment.” So, when a patient has particular attractor features that associate with sepsis, for example, this could trigger an alert to the clinical team.
The preclinical tests have shown promising results, suggesting clear differences in the attractor between a healthy subject and one in the early stages of sepsis – but this has to be validated in humans.
Manasi explains, “what's interesting is if you just look at the conventional waveform measures of blood pressure, you wouldn't necessarily know that there was anything wrong. The attractor seems to be very sensitive at detecting something that happens in those really early stages of sepsis, once that microbial infection is taking hold. Something happens in the cardiovascular system and it seems that we're able to sensitively detect that with our method. In the conventional signal, it only becomes obvious about four or five hours later.”
One of the reasons for this is that the human body has evolved to divert blood from certain body systems, such as the gut and skin, in order to maintain blood pressure to vital organs such as the heart and brain when the body goes into ‘shock’. This can mean that a conventional signal like measured blood pressure can appear normal even though systems have started to close down in the body and the patient is actually getting ill.
Manasi’s initial research has shown that the attractor method is very sensitive at detecting cardiovascular changes in the earliest stages of sepsis compared to conventional cardiovascular measures such as systolic/diastolic blood pressure or heart rate.
It’s this early warning system that has the potential to save lives in settings ranging from an ICU to the Accident and Emergency Department.
Manasi says, “I'm really excited to be working on this project because it has immediate potential in the clinic. With a lot of the research that I've done to date you're 10, 15, 20 years away from it ever being tested in patients. Whereas something like this we're starting to test the system within 5 years - that's really exciting.”
“In terms of how that would impact on healthcare as a whole? If we can diagnose these patients earlier, they've got a much greater chance of surviving and leaving hospital. And the earlier you do it, the less likely they are to have ongoing complications. There's the benefit to the patient and their families. But there is also a huge benefit to the healthcare system because it means that those patients are not going to be hospitalized for as long. There are both economic and societal benefits of an early detection method.”
Manasi says, “attractor reconstruction has very wide application that is above and beyond recognition of patients with sepsis. But we're applying it to sepsis in the first instance because that’s a really big clinical problem, and it's one where patients are already routinely monitored using continuous bedside monitors – so we have the data we need.”
Attractor reconstruction has the potential to be extended to predicting a range of health conditions and could even be used to help understand how effective drugs are, or whether they carry a safety risk.
Manasi says, “We are in the earliest stages, but the project is getting into quite an interesting space at the moment. We’re collaborating with lots of different doctors and research scientists and they're sending us their data and we are starting to see if this method can be applied to other areas.”
Using LabChart to extract high-fidelity waveforms
Manasi uses ADInstruments’ LabChart software to extract the high fidelity cardiovascular waveforms that are used to build the attractor. The waveforms can come from blood pressure, ECG, or pulse oximetry data. She says, "I use LabChart for a variety of applications in my work. The software allows you to take averages of routine signals like systolic and diastolic pressure easily. But at the same time, in the background, it's collecting really high-fidelity data and sampling it at a thousand data points per second or a thousand Hertz. It means that I can go ahead and extract that big data out of LabChart and feed it into our new bit of coding, which then transforms it into an attractor. So LabChart allows me to do both a conventional analysis but also gives me the raw data to feed into the new system to do the new analysis.”
“When we look at our human volunteers, we often rig them up with lots of different devices on them. So, we'll be measuring a fingertip blood pressure reading, we'll also be getting ECG monitoring from them at the same time. And it means we've got a continuous stream of data from that individual that tells us a lot about their cardiovascular system.”
The importance of collaboration: where maths meets medicine
For Manasi, collaboration and interdisciplinary research is hugely important. “Philip and I have grown our team and have brought in mathematicians and biologists. But I also get to work with bioengineers, with statisticians, with doctors, with nurses. You get to find out everyone's take on a problem and you also get to see different ways in which they think about things and you learn from each other. I really enjoy that aspect of the job. And by working together we're able to answer some really interesting and important scientific problems.”
“I think what is great is that we can apply our different ways of thinking to solve complex problems together - and it’s been really liberating because there are no set rules about how to do it. With the mathematicians it was fascinating to see them at play with the data until things started to make sense or until we could say "that's really novel, that's not been done before, that could be really important".
“It's a really great, interdisciplinary project and I’ve had to learn this other “language”. Most of my time is spent communicating maths to biologists or communicating the biology to mathematicians. It’s been a really steep learning curve for me and I’ve realized that mathematics is a very pure science - it's just taking what it is given. With biology we tend to apply our interpretation to what bit means what. By taking a mathematician’s approach, and just looking at everything we've been able to discover this new information.”
“I think what's really great about doing this type of research is that you can have an impact - in this case, on human health. I'm really excited that this project is now a stage where we're starting to work with clinical data. It takes a lot of work to get to this point. And to know that this project has a potential to be implemented in clinical practice as an early warning system... to me that is incredibly exciting.”
While Manasi finds the current project exciting, she originally never envisaged working so closely with mathematics.
She says, “it's strange when you're an undergraduate and you don't really know where you're going to end up in 10 or 15 or 20 years’ time. If you told me I was going to be working on a maths and physiology project I wouldn't have believed you ten years ago! I was very much focused on in vivo, cell and molecular biology based research and working in the laboratory rather than being a data scientist. But I definitely knew I wanted to work to do something associated with healthcare research.”
Manasi is a pharmacologist, focusing on the cardiovascular system. She says, “a lot of my interest and research to date has been around trying to identify new drug targets to treat certain cardiovascular diseases once they've been established.”
Manasi completed her PhD at the Institute of Child Health and postdoctoral training at University College London in the laboratory of Professor Patrick Vallance. During this time, she developed a number of in vivo systems to characterize a novel mouse mutant and small molecule, and identified a nitric oxide modifying pathway as a novel target for the treatment of septic shock. She was awarded a British Heart Foundation Intermediate Fellowship immediately prior to commencing her lectureship at King’s College London in 2009.
On a daily basis, her work is varied. “I still do some wet lab stuff. For example, this week I did some surgery, implanting small blood pressure monitoring devices in animal models to try and extract experimental data for my colleague. A lot of my time is spent in front of a computer looking at data sets which collaborators have sent us, and there's a lot of meetings with the mathematicians.”
“Research is a really stimulating job. I meet incredibly interesting people and form collaborations with them. You're constantly inspired and you're constantly learning, and I'd say to anyone who's not sure about their career options - make sure you do something that interests you.”
“One thing leads to another in research and that's what I really like - that you can be working on quite a niche, focused project for quite a long period of time but you can't necessarily see the bigger picture and where it’s leading. And then, before you know it you branch off on something else.
“Before you know it, your project will end up branching off in different directions and if you follow some of those, they can lead you to really interesting places. So, I’d say to anyone starting out in research to take risks and go outside your comfort zone but also make sure you're interested in and inspired by what you're doing. The job is really, really stimulating - no two days are the same and that's probably what I like the most about being a researcher.”
- 1: http://ehealth.kcl.ac.uk/cardiomorph/
- 2: CDC - https://www.cdc.gov/sepsis/index.html
- 3: https://www.sepsis.org/downloads/2016_sepsis_facts_media%20copy.pdf
- 4: https://www.ncbi.nlm.nih.gov/books/NBK65391/
- 5: https://www.theguardian.com/lifeandstyle/2017/sep/18/sepsis-the-truth-about-this-hidden-killer
- 6: https://www.bmj.com/content/350/bmj.h3017
- 7: Recknagel P, et al. (2012). Liver Dysfunction and Phosphatidylinositol-3-Kinase Signalling in Early Sepsis: Experimental Studies in Rodent Models of Peritonitis. Retrieved Aug 22, 2018 from http://www.plosmedicine.org/article/info%3Adoi%2F10.1371%2Fjournal.pmed.1001338
- 8: Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, Sharma S, Suppes R, Feinstein D, Zanotti S, Taiberg L, Gurka D, Kumar A, Cheang M. (2006) Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 34(6): 1589-96. DOI: 10.1097/01. CCM.0000217961.75225.E9
- 9: http://ehealth.kcl.ac.uk/cardiomorph/
- 10: http://ehealth.kcl.ac.uk/cardiomorph/downloads/2018-02/beyond-hrv.pdf
