
In human autonomic studies it is crucial to create a setting that ensures you can acquire reliable and accurate data.
Dr Jackie Limberg, Assistant Professor at the University of Missouri, shares ten easy ways to anticipate and avoid situations that may increase data variability and which may impact your ability to interpret the results.
Preparing Your Lab
#1: Quiet
We all need some peace and quiet...and so do your research participants. This might seem trivial, but it is surprising how even a slight noise or distraction in the room can affect your data. Even the opening of a drawer or noises from the next room can cause changes in basal autonomic activity found at rest.
#2: Temperature
In autonomic research we are measuring changes that the participants are not consciously thinking about, including their involuntary responses to a room that is slightly too hot or too cold. To avoid this, keep your room at a constant and moderate temperature. The ideal temperature range is between 21 - 24°C ( 69.8 - 75.2° Fahrenheit).
#3: Lighting & Timing
Autonomic function differs between the hours of wakefulness and sleep.
If your perfectly quiet room at the ideal temperature is also dimly lit, your participants may start to doze off. So if your research question requires participants to be fully awake, be sure that your lab has sufficient lighting. It is also advisable that the session is scheduled during the day, around the same time for each repeat experiment!

Photo by Kelly Sikkema on Unsplash
Before Your Study
#4: Activites
Make sure your participants refrain from alcohol, caffeine, intense exercise, and smoking/nicotine for at least 12 hours before an experiment. Alcohol and caffeine intake have both been shown to increase MSNA activity .
In addition, eating a large meal has been shown to increase MSNA for up to 90 minutes afterwards. Participants should fast for at least 3 hours prior to the study. (Hart et al., 2017).
#5: Medical History
It is standard practice to inquire about past and current medical history and current medications to determine whether participants meet eligibility criteria. Medications that are known to impact the autonomic, cardiovascular or endocrine systems should be documented. These include oral contraceptives and hormone replacement therapy.
Menstrual cycles can also exhibit strong effects on autonomic function. Click here for detailed information guides on how to control these effects during your study.
During Your Study
#6: Micturation
The need to use the restroom can elicit similar physiological responses to pain and discomfort, which can notably increase sympathetic nerve activity.
Surprisingly, these effects can be observed up to 30 minutes before the bladder empties (micturition). If your participant says they need to use the restroom, keep in mind that your data may have been compromised for the previous half an hour!
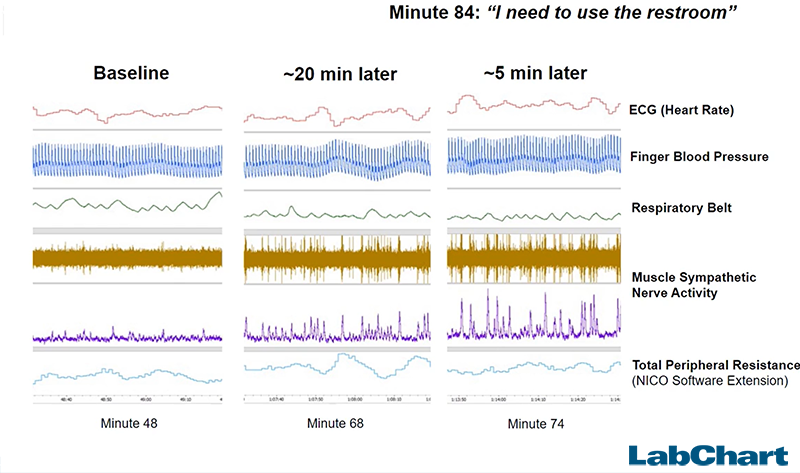
#7: Having a Rest
It is recommended when measuring autonomic nervous system variables that you allow at least 20 minutes for a quiet resting period once your participants arrive at the lab. This will reduce the effects of any stress associated with travelling to the lab, whether it’s getting lost in the hallway or running late!
#8: No Talking
When the participant is talking, you are no longer measuring their basal autonomic control. You are measuring changes in the heart rate or muscle sympathetic nerve activity (MSNA) in response to somebody talking. Therefore if this happens, the testing period should be repeated where possible.
TIP! A comfortable respiratory belt is an effective way to ensure changes you are seeing are true and not just an artifact of participants moving or talking. Matching the data from the respiratory belt to your dependent variables, such as heart rate or blood pressure, will flag up any data anomalies attributed to moving or talking. Jackie uses the Respiratory belt transducer to increase the validity of her data.
Your HRV Data
#9: Sampling rate
It is crucial that the sampling rate is at least 500 Hertz. When measuring heart rate variability (HRV), the difference between the R-R interval of each heart beat could be less than a millisecond. If the sampling rate is too low, it can be impossible to detect these differences.
The BioAmp digital filter can be very useful when collecting ECG data. However, if you are using an external filter, she recommends that you check and understand what the filtering is on the device before drawing conclusions from your HRV data.
#10: Consistent Data
When measuring HRV, the minimum required file length is about five minutes (~ 250 cardiac cycles) - go any shorter and you risk not capturing very low frequency HRV changes. The filing that you use needs to be consistent between participants and interventions because the normalized units (NU) take into account total power of the file. This will vary significantly based on file length and HR of the participant.
For consistency, it is also best practice to go through the recordings and manually correct recordings of ectopic beats or arrhythmias; these will distort the R-R intervals and decrease the validity of analysis assumptions.
RESOURCE: Investigators should/should not control for the menstrual cycle phase when performing studies of vascular control that include women.
https://pubmed.ncbi.nlm.nih.gov/32702264/ (Point)
https://pubmed.ncbi.nlm.nih.gov/32702274/ (Counterpoint)
https://pubmed.ncbi.nlm.nih.gov/32702278/ (Rebuttal)
https://pubmed.ncbi.nlm.nih.gov/33197373/ (Last Word - Point)
https://pubmed.ncbi.nlm.nih.gov/33197375/ (Last Word - Counterpoint)
https://pubmed.ncbi.nlm.nih.gov/33197376/ (Commentaries)
We hope this information was useful to you and we thank Jackie for the generosity of sharing her time and knowledge.
You can view the full webinar here.

Jacqueline Limberg, PhD
Assistant Professor, Department of Nutrition & Exercise Physiology
University of Missouri
Dr. Limberg is a human integrative physiologist with a primary focus on the role of the autonomic nervous system in cardiovascular responses to exercise and environmental stressors in health and disease.
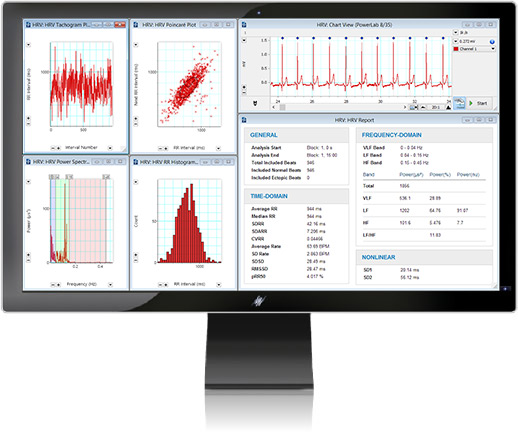
LabChart - HRV for Human Autonomic Studies
With the Heart Rate Variability (HRV) Module in LabChart you can perform robust beat detection, power spectral analyses and creates tachograms, histograms, Poincaré plots and statistical reports. You can even automatically or manually exclude ectopic beats with the Beat Classifier view. Contact us for more information.
