Are you thinking about using optogenetics in your neuroscience research but are unsure where to begin? Here we provide an overview of the key tools and techniques involved in optogenetics and its applications in the neuroscience field.
Overview
What is Optogenetics?
Optogenetics is a relatively new technique in the neurobiology field that integrates the use of light (optics) and genetic engineering to manipulate neural activity. It broadly involves transfecting neurons with light-sensitive proteins (opsins) that, depending on the type of opsin used, can selectively activate or silence those neurons using pulses of visible light.
Optogenetics has now taken hold in the neuroscience field and revolutionized how we study the brain. Prior to its application, it was challenging for scientists to discriminate the neural pathways involved in certain physiological responses and behaviors. Alternative methods such as pharmacological or electrical stimulation lack the same temporal and anatomical precision that can be achieved through optogenetic tools.
With the rapid development of related technologies, optogenetics has opened the door to therapeutic applications such as deep brain stimulation for the treatment of Parkinson’s disease, prevention of seizures for epilepsy, and even in the pacing of heart muscle for cardiac dysfunction.
Optogenetics offers a highly targeted approach to neural modulation from the scale of individual neurons in living tissue, to freely moving animals - making it a powerful tool for any neuroscience researcher.
So how does it work?
Although a wide range of technologies now fall under the optogenetics umbrella, generally they can be divided into two main categories; the tools used to express opsins in the target tissue, and those used to stimulate (activate/deactivate cells) and measure the resulting response.
This video provides an overview of some of the tools and techniques used for opsin expression and stimulation for in vivo animal studies, which we will go into more detail below.
Opsin expression in the target tissue
Because microbial opsins aren’t naturally expressed in mammalian tissue, the target cells must be genetically engineered to express these proteins.
Several techniques are available for introducing opsins into live cells, including DNA transfection, electroporation, and transgenic expression systems. In the brain, viral-mediated gene delivery has become the method of choice for most neuroscientists due to its ability to target different regions of brain tissue with increased accuracy.
There are three main components to a viral expression system:
- The Opsin - light-sensitive protein
- The Promoter - used to target specific cell types
- The Reporter - Used to detect and quantify the amount of protein expression e.g Green fluorescent protein (GFP)
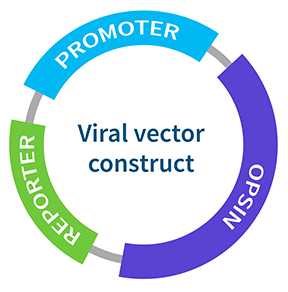
Each of these components can be tailored to suit the specific research question. For example, there are hundreds of different types of opsin variants available, each with their own unique spectral and kinetic features that can be used to target different cell types and elicit different physiological responses (Figure 1).
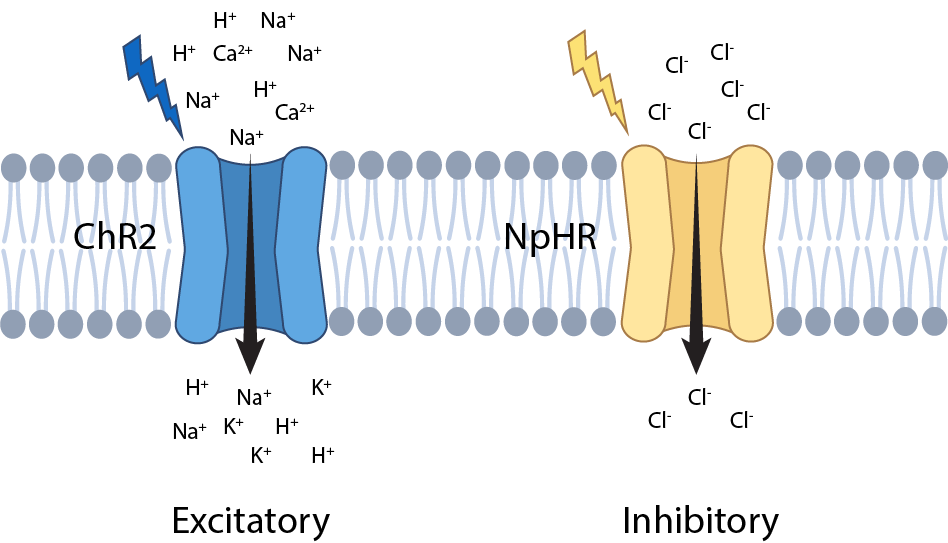
Figure 1. Examples of different opsin variants. Upon exposure to blue light, Channelrhodopsins 2 (ChR2) allows cations to enter the cell causing an excitatory response. In contrast, Halorhodopsin (NpHR) upon exposure to yellow light, results in an inhibitory response.
For instance, channelrhodopsins are a commonly used ‘excitatory' variant of opsin that activate cells in response to blue light. Whereas halorhodopsins are an inhibitory type of opsin that in response to yellow light, allow negative ions to flood into the cell, inhibiting neuron activity.
Likewise, there are various viral expression systems available in the optogenetics toolkit, each designed to work in different animal models or facilitate different types of opsin expression.
The Adeno-Associated Virus (AAV) is the most widely used viral vector in small animal research due to its specificity for infecting neural tissue and relatively low inflammatory rates compared to other viral vectors.
Once the viral vector has been constructed and amplified, microinjection is generally used to deliver the virus to the target region in the brain. The virus will then infect localized neurons and deliver the construct. A chosen subset of those neurons will have the right cellular machinery to activate the promoter and express the opsin in the cell.
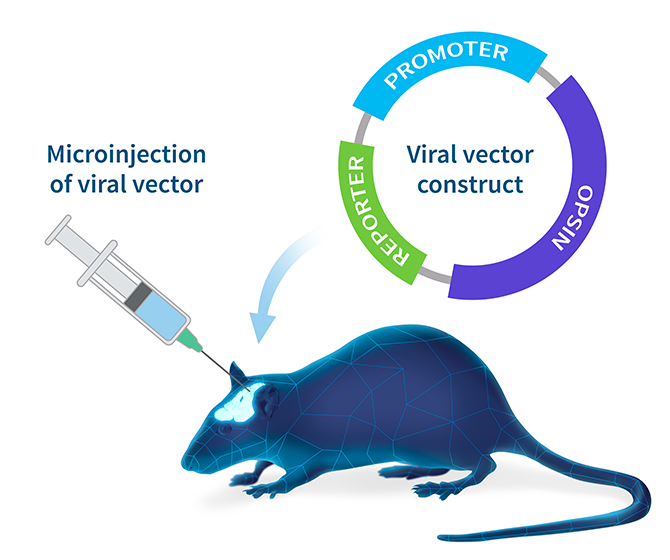
For more detailed information about viral vector expression systems and the steps involved in creating and isolating a viral vector for optogenetic studies, check out this helpful guide.
Light stimulation of opsins expressing cells
The second major factor that needs to be considered for optogenetic studies are the tools required to stimulate the opsin expressing cells and measure the resulting response. Most light targeting devices available for in vivo animal studies involve the use of a tethered or wireless head-mounted system, with the addition of a secondary piece of equipment for recording the physiological response.
While tethered and head-mounted systems have improved dramatically since the initial application of optogenetics, they still have their limitations, including:
- Increased susceptibility to infection at interface site
- Impact on normal physiology and behavior (due to tethering or bulky headpiece)
- Restriction of experimental duration and protocols due to restraint and/or limited battery-life
- Additional costs of tether maintenance and replacement
Telemetry-based light stimulation
With optogenetics now taking a shift towards therapeutic research applications, there is a need for light delivery/stimulation tools that are more suited to long-term studies. As such, wireless telemetry-based systems are becoming increasingly popular for in vivo animal research, as they facilitate long-term, unrestrained experimentation on animals in their home environment.
Related: Benefits of using telemetry for small animal research »
Introducing the Kaha Sciences Optogenetics telemeter
The Kaha Optogenetics telemeter is the first fully implantable wireless optogenetic solution to offer both optogenetic stimulation and recording (EEG, ECG, and EMG) within the same device. Designed for use in rats, the telemeter is an ideal solution for researchers looking to apply extended optogenetic stimulation protocols and record quantitative biopotential data in long-term experiments without the restrictions of battery power and tethers.
Kaha Sciences Optogenetics Telemeter (TR58AB)
For more information about the Kaha Optogenetics telemeter and how it might be useful for your particular research application, please get in touch with your nearest ADInstruments representative.
Please note that ADInstruments is unable to support customers in the development of their viral vectors for opsin expression. If you are new to optogenetics, we suggest collaborating with a researcher who is experienced in the techniques required to create a viral vector expression system.
Helpful Resources:
Optogenetics: 10 years of microbial opsins in neuroscience. Deisseroth, K. (2015). Nat Neurosci 18, 1213–1225. https://doi.org/10.1038/nn.4091
A Hitchhiker’s Guide to the Selection of Viral Vectors for Optogenetic Studies. Thompson K.R., Towne C. (2018). In: Stroh A. (eds) Optogenetics: A Roadmap. Neuromethods, vol 133. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-7417-7_1
Making Sense of Optogenetics. Guru, A., Post, R. J., Ho, Y. Y., & Warden, M. R. (2015). The international journal of neuropsychopharmacology, 18(11), pyv079. https://doi.org/10.1093/ijnp/pyv079
The development and application of optogenetics. Fenno, L., Yizhar, O., & Deisseroth, K. (2011). Annual review of neuroscience, 34, 389–412. https://doi.org/10.1146/annurev-neuro-061010-113817
Optogenetics: controlling cell function with light. Pastrana, E. (2011). Nat Methods 8, 24–25. https://doi.org/10.1038/nmeth.f.323

