Customer Snapshot - Dr Fiona McBryde
Overview
Fiona McBryde is a Cardiovascular Physiologist and Senior Lecturer at the University of Auckland, whose pre-clinical research focuses on understanding blood pressure regulation in the brain in response to life-threatening diseases like ischemic stroke.
Through the use of small animal telemetry, she is able to perform long-term studies looking into the physiology of stroke and how signals like blood pressure, brain tissue oxygen, and SNA activity change in response to an ischemic attack.
Fiona’s research is unique as it extends beyond the lab and into the clinic, where she collaborates with clinicians at a local hospital to better apply her research to clinical practice - with the ultimate goal of improving stroke-patient management and outcomes.
‘We're trying to help shine some light on what might be the best practice to guide clinicians. They can't run a clinical trial for every variable - so we're trying to narrow down the options to the ones most likely to have a benefit.’ - Fiona McBryde, Senior Lecturer, University of Auckland.
They say time is brain when it comes to strokes.
A stroke can be likened to a heart attack, but in the brain - a sudden interruption of blood flow to our most precious organ, and one of the leading causes of mortality and disability worldwide 1,2.
Approximately 87% of strokes are ischemic2, caused by a clot or fatty build-up (plaque) in an artery of the brain. Within minutes of an ischemic stroke occurring, brain cells (neurons) begin to die due to a lack of oxygen.
To treat an ischemic stroke, immediate medical intervention is critical. However, even for those fortunate enough to recognize the signs of stroke early and get to a hospital, there is currently no ‘silver bullet’ treatment.
For more than 20 years, the only proven treatment for ischemic stroke was intravenous thrombolysis (IVT) - a medication used to dissolve the clot in-situ. However, ~40% of ischemic strokes are caused by large artery blockages which are often unresponsive to thrombolysis treatment.
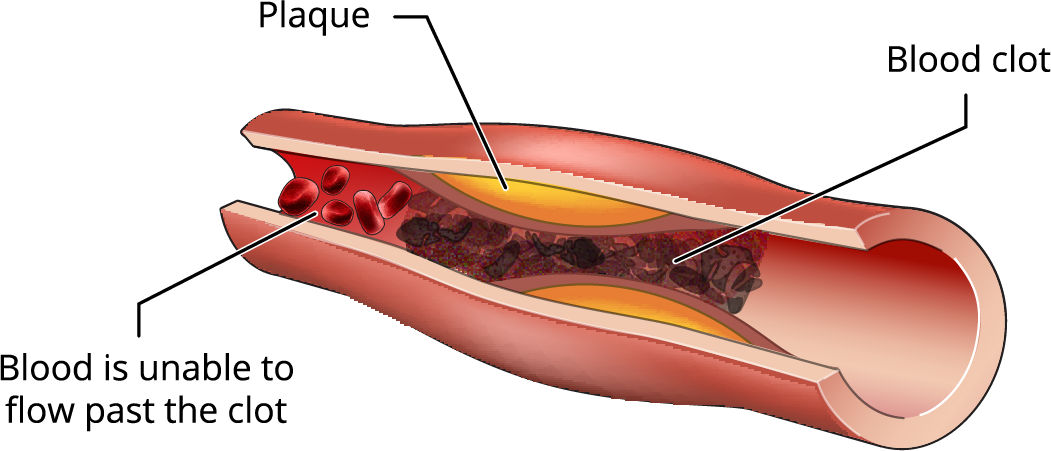
Ischemic strokes occur when the blood supply to the brain has been restricted in some way.
The Endovascular Thrombectomy - A life-saving procedure
In 2008 however, there was a significant breakthrough in the treatment of these large artery occlusions, with the development of a surgical procedure called an Endovascular Thrombectomy (EVT). Instead of relying on medication to dissolve the clot, EVT involves mechanical clot disruption to achieve the removal of the clot3,4.
‘The EVT has had a massive impact on patient outcomes. Previously, many stroke patients were considered beyond help. Treatment plans were focused on rehabilitation and trying to gain back lost brain function. Now, they have the option to go in and actually remove the clot and start rescuing brain tissue before the real damage is done,’ explains Dr Fiona McyBryde.
The procedure involves threading a catheter up through the femoral artery in the groin, past the heart, and into the blocked artery in the brain. A stent on the end of the catheter then opens and retrieves the clot, allowing blood flow to return to the injured tissue.
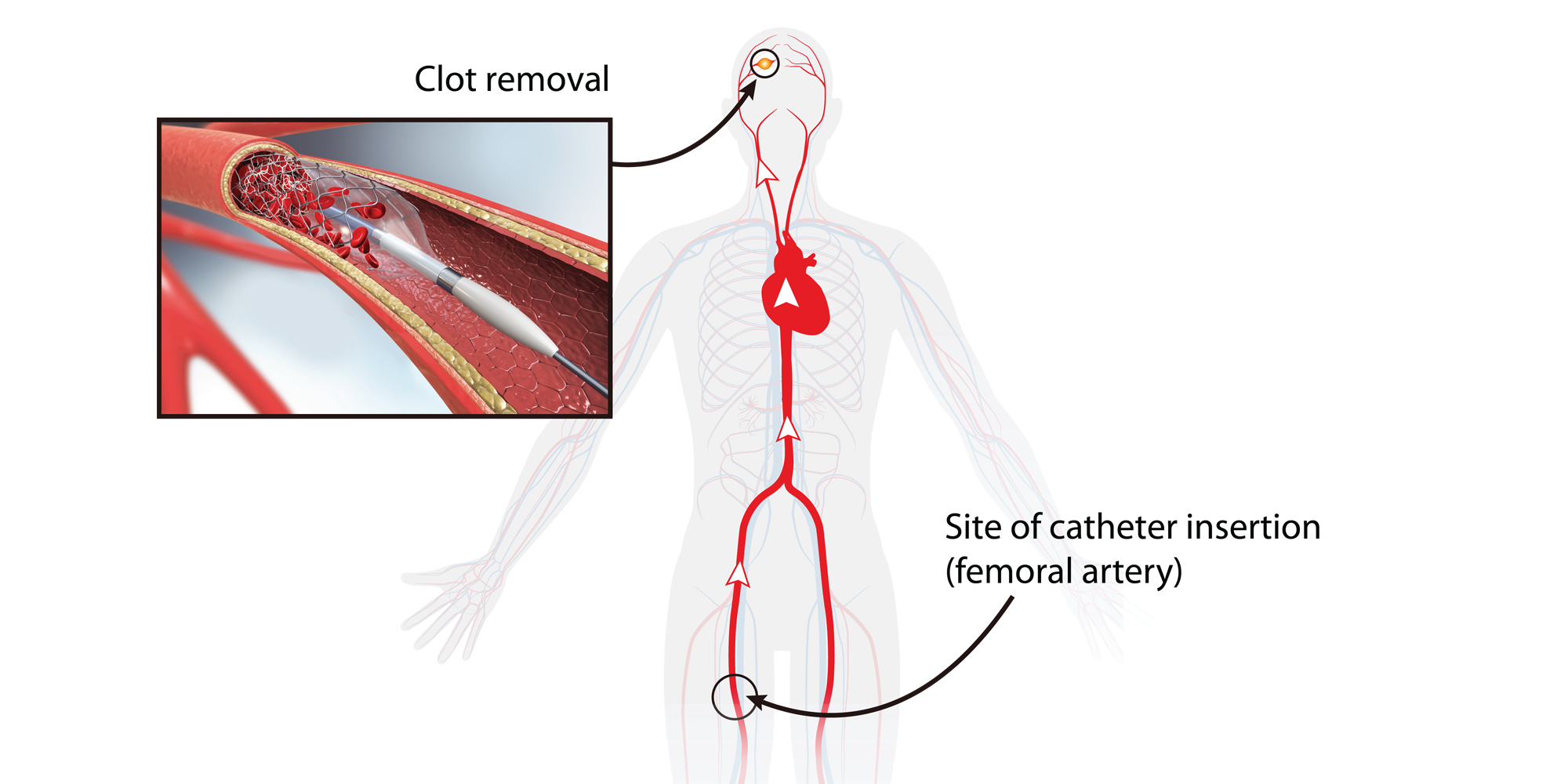
Improving stroke patient outcomes through blood pressure modulation
Although the introduction of the EVT has been a significant leap forward in terms of patient survival and recovery, the procedure itself still presents a variety of unanswered questions regarding the management of stroke patients during and after the surgery.
Fiona explains, ‘In New Zealand, clot retrieval is performed under general anesthesia. However, when you have an anesthetic, the body's normal homeostasis pathways are affected. As a result, the patient’s blood pressure tends to fall.’
This means clinicians have to decide where to set that patient's blood pressure during the procedure. Currently, an approach of ‘one blood pressure fits all’ is generally used in surgeries requiring general anesthesia. The problem with this is that it assumes all patients are going to respond in the same way.
However, stroke patients are unique as many (up to 85%3) suffer from chronic hypertension (high blood pressure), prior to stroke occurring. While many of these patients will be on medication to combat their hypertension, there is a portion who have undiagnosed hypertension. This extra level of complexity introduces additional uncertainty when deciding where to set the patient's blood pressure during the procedure.
‘It’s a big problem, and one that’s hard to address in the clinical setting’, says Fiona. ‘The clinicians attending those stroke patients have no way of knowing if blood pressure is coming up and down, and what effect that's having in the brain. It's a bit of a black box.’
‘You don't want to set the blood pressure too high as you risk having complications like bleeding on the brain or the bursting of aneurysms. However, there’s also a real danger in setting it too low, because you risk not getting enough blood flow through to those already vulnerable areas, which ultimately means there'll be less tissue to rescue when you eventually get the blood clot out.’
‘What I'm trying to do with my pre-clinical studies is unpack that black box and see what's actually happening to blood vessels in the brain during stroke. And what we really want to understand is whether we can help improve blood flow to that really vulnerable tissue in the brain by modulating blood pressure.’

‘They say time is brain when you've got an occlusion, you're losing millions of neurons and neuronal connections every minute that occlusion goes untreated. So it's quite a short window we have to work within, but actually quite a profound potential for benefit to the brain if we can get this right.’
Understanding the physiology of stroke using telemetry
In order to delve deeper and understand what is happening in the brain during stroke, Fiona has developed an animal model of ischemic stroke, and uses small animal telemetry devices to measure the physiological response. Some key signals they are interested in measuring are blood pressure, brain tissue oxygen concentration, intracranial pressure, and sympathetic nerve activity.
Fiona explains, ‘We've got telemetry devices that let us measure these physiological signals for long periods of time. The important thing is that we are able to measure them in animals that are just going about their normal daily life. We don't want to be stressing them out, we don't want to be handling them any more than we have to, and we just want to be able to measure these signals remotely - and the telemeters allow us to do that. ’
Find out more: Exploring ICP, Tissue Oxygenation and RSNA with Implantable Telemetry »
The selfish brain hypothesis
Recently, Fiona and her team have published some papers that look into what happens to blood pressure after stroke i.e. prior to any medical intervention. 'For these studies, we wanted to understand if the body's response varies between those with normal blood pressure, those with high blood pressure, or individuals with high blood pressure that are receiving chronic treatment - as each of these scenarios are possible in the real world,' says Fiona.
‘Interestingly, what we saw was that regardless of where your blood pressure was sitting prior to stroke, the cardiovascular response was virtually identical,' says Fiona.
‘You get this big surge in blood pressure and powerful activation of the nervous system for several days after the stroke occurring - a response we call the selfish brain hypothesis. We believe that the brain, through its control of the autonomic nervous system, has the ability to modulate systemic blood pressure. And if something happens to threaten blood flow to the brain, like stroke, the brain has the capability to actually demand hypertension for the rest of the body in order to sustain its own perfusion.’
The future of stroke research
More recently, Fiona and her team have performed studies looking into changes in intracranial pressure and brain tissue oxygen concentration before and after ischemic stroke, as well as the relationship between blood pressure and blood flow in the brain.
‘My projects tend to be fairly ambitious,' says Fiona. I like to record a lot of signals, whether that's using one or combinations of different telemeters i.e. two in one animal.'
‘I guess I have a toolbox approach to science. I see the telemeters that are available, and the signals that we can look at, and combine these together with our specialist techniques in different ways to ask some quite intricate and unique questions about the physiology.’
Even though Fiona's experiments are still in the pre-clinical stage, her findings will play an important role in deciding the direction of clinical trials in the future. ‘Ultimately, we're trying to help shine some light on what might be the best practice to guide clinicians. They can't run a clinical trial for every variable - so we're trying to narrow down the options to the ones most likely to have a benefit.'
ADInstruments would like to thank Fiona for taking the time to share her lab's cutting-edge research with us. For more information about her current projects, you check out Fiona's most recent publications here.
Telemetry for Cardiovascular Research
Take your cardiovascular research to the next level using small animal telemetry - with options to measure pressure, ECG, sympathetic nerve activity, and tissue oxygen concentration, continuously over the course of your experiment.
Related resources:
More about Kaha Sciences Small Animal Telemetry »
Tissue oxygen telemetry: How does it work and why is it beneficial to your research? »
Kaha Telemeters Cardiovascular Applications »
Four benefits of telemetry for research »
How to measure chronic intracranial pressure in rats »
References:
1. World Health Organisation 2020, The top 10 causes of death, The United Nations, accessed 1 August 2021, <https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death>.
2. Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, et al. Heart disease and stroke statistics—2020 update: a report from the American Heart Association. Circulation. 2020;141(9):e139–e596.
3. Papanagiotou P, Roth C, Walter S, Behnke S, Politi M, Fassbender K, Haass A, Reith W. Treatment of acute cerebral artery occlusion with a fully recoverable intracranial stent: a new technique.Circulation. 2010; 121:2605–2606. doi: 10.1161/CIRCULATIONAHA.110.948166.
4. Pérez MA, Miloslavski E, Fischer S, Bäzner H, Henkes H. Intracranial thrombectomy using the Solitaire stent: a historical vignette.J Neurointerv Surg. 2012; 4:e32. doi: 10.1136/neurintsurg-2011-010149.
5. McManus M, Liebeskind DS. Blood Pressure in Acute Ischemic Stroke. J Clin Neurol. 2016;12(2):137-146. doi:10.3988/jcn.2016.12.2.137


