While Millar pressure catheters are primarily designed for measuring highly accurate cardiovascular pressures, these sensors have been used for measuring a range of other physiological pressures within the body such as tumor interstitial fluid, airway, and spinal (intradiscal) pressures.
Here we discuss some of the less well-known applications of the pressure catheter including, why and how to measure pressure in these different areas, plus helpful resources to get you started!
#1 Tumor Interstitial Fluid Pressure
The fluid within the body can be conceptually divided into three main fluid compartments; intravascular, interstitial, and intracellular.
![]()
The interstitial fluid, located between tissue cells, makes up approximately one-sixth of the body volume and plays a vital role in the exchange of oxygen, nutrients, and waste products between the vascular system and cells.
Movement of water and molecules across these compartments, also known as transcapillary flow, is mediated through hydrostatic and osmotic forces (or pressure gradients), imposed by the fluid and proteins within the capillaries and the surrounding interstitium.1
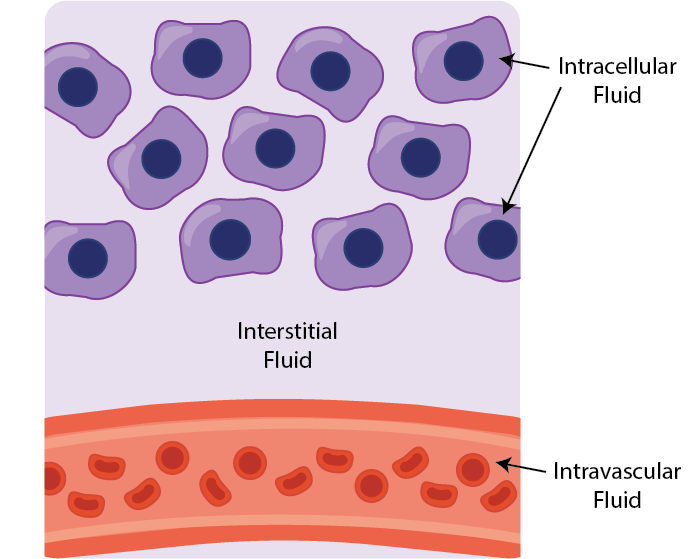
There are three major fluid compartments in the body; intravascular, interstitial, and intracellular.
Normally, interstitial fluid pressure (IFP) is regulated through the interactions of stromal cells and the extracellular matrix molecules. However, often within solid tumors, the IFP is significantly higher than normal, forming a barrier against efficient drug delivery into the tumor. 1 Understanding how this increase in IFP affects the penetration of drugs into various tumors is a fundamental feature of cancer biology.
By monitoring tumor IFP in vivo, researchers have been able to observe a correlation between tumor growth, increased IFP, and reduced treatment response. Mice are by far the most commonly used animal model in cancer research, leading to a range of well-established methods for direct and indirect measurement of tumor IFP. Direct methods measure the magnitude of the pressure. In contrast, indirect methods measure a quantitative value that can be used to derive the tumor pressure through a computational algorithm or scaling factor. 2
Below we will take a look into the direct measurement of IFP for cancer research.
How is Tumor IFP measured?
Typically, tumor IFP can be measured by inserting a measurement probe directly into the tumor tissue. The two most commonly used methods for measuring tumor pressure are the wick-in-needle and pressure catheter methods.
The pressure catheter method, however, has the added benefit of detecting both IFP and solid stress; an important factor for determining the exact IFP of tumors.2
It involves inserting a calibrated pressure catheter into the tumor of the anesthetized animal. Often a guide needle is used to penetrate the tumor or tissue of interest first; then, the catheter probe is inserted once the needle has been removed.
Related: How to calibrate your Millar Pressure Catheter »
The catheter is then connected to a Bridge Amplifier and PowerLab data acquisition system options to acquire a pressure signal. The pressure signal can then be visualized and analyzed within LabChart software.
Millar catheters provided a simple, accurate method for recording tumor interstitial fluid pressures in a variety of models. Millar's SPR-1000, SPR-320, and SPR-524 pressure catheters have all been used to deliver high-quality tumor IFP measurements (see publications below).
Publications citing Millar technology for IFP measurement:
- A simple method for measuring interstitial fluid pressure in cancer tissues (2005) - SPR-320 catheter
- Enzymatic Targeting of the Stroma Ablates Physical Barriers to Treatment of Pancreatic Ductal Adenocarcinoma - SPR-1000 catheter
- Antifibrotic Therapy Disrupts Stromal Barriers and Modulates the Immune Landscape in Pancreatic Ductal Adenocarcinoma (2019) - SPR-1000 catheter
- Interstitial Pressure in Pancreatic Ductal Adenocarcinoma Is Dominated by a Gel-Fluid Phase (2016) - SPR-1000 or SPR-671 catheter
#2 Transdiaphragmatic Pressure
The diaphragm, located just below the lungs and the heart, plays a vital role in the breathing process, as well as other behaviors required for airway clearance such as coughing, vomiting, and sneezing. It aids in ventilation and non-ventilatory behaviors by producing varying levels of force (through muscle contraction and relaxation), which can be defined by measuring the transdiaphragmatic pressure.3
Transdiaphragmatic pressure is particularly important when understanding how injury and diseases affecting the diaphragm (e.g., neuromuscular disorders) can impact on its ability to generate force for its various functions.
Studies have measured it in a range of species, including humans (clinically), sheep, dogs, rabbits, rats, and mice.
Traditionally, in larger subjects, transdiaphragmatic pressure is measured using a dual balloon catheter system, where balloon-tipped catheters are inserted through the nose into the esophagus and the stomach, in order to measure esophageal pressure (Peso) and gastric pressure (Pgas) respectively.4 The transdiaphragmatic pressure can then be derived from the difference between these two pressure signals.
Recently, studies have adapted the use of solid-state pressure catheters for this technique, which is particularly useful in smaller animals such as mice, where insertion of the balloons can be difficult.3
How is transdiaphragmatic pressure measured?
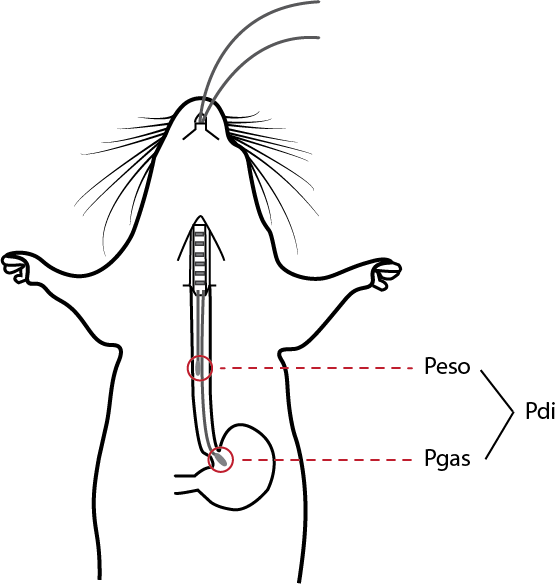
Schematic for measuring transdiaphragmatic pressure (Pdi) in mice.
For this technique, two pressure catheters (e.g., a 3.5 F, SPR-524 Millar catheter) are inserted through the esophagus of an anesthetized mouse, one into the stomach to measure gastric pressure (Pgas) and the other in the esophagus to measure the thoracic esophageal pressure (Peso). The signals from the catheters pass through a Bridge Amplifier, connected to a PowerLab data acquisition system. The pressure signals are analyzed within LabChart, and the transdiaphragmatic pressure can be automatically calculated from the difference between Pgas and Peso.
Note that in addition, depending on the animal species, size, and spacing required between the two pressure sites, two rat dual pressure catheters 2.5F, SPR-721 & 2.5F, SPR-940 are also available.
Check out the publications below for more detailed information on how to measure transdiaphragmatic pressure using Millar pressure catheters.
Publications citing Millar technology (SPR-524 catheter) for transdiaphragmatic pressure measurement:
- Functional Measurement of Respiratory Muscle Motor Behaviors using Transdiaphragmatic Pressure (2017)
- Novel method for transdiaphragmatic pressure measurements in mice (2013)
- Functional impact of diaphragm muscle sarcopenia in both male and female mice (2015)
- Impact of Unilateral Denervation on Transdiaphragmatic Pressure (2016)
#3 Spinal or Intradiscal Pressure
The spine, quite literally, is the backbone of your body - without it, you wouldn't be able to do simple movements such as walking, bending over, or picking up objects. Another important function of the spine is protecting the spinal cord and the associated nerve roots, allowing your brain and the rest of your body to communicate with each other.

Measuring intradiscal stresses or pressures can give you information about various loads acting across the spine. This can be particularly important when studying conditions such as neurodegenerative diseases, scoliosis, disc herniation, or spinal cord injury, which affect how various stresses or loads are distributed across the spine. Animal models, such as sheep, are particularly useful in this field as the shape of their spines are similar to humans.
How is intradiscal pressure measured?
Intradiscal pressure can be monitored by inserting a pressure transducer into the nucleus pulposus of an intervertebral disc, either directly during an open discectomy 5 or using a protective spinal needle with a suitable Millar pressure transducer inside (as shown on the right).6
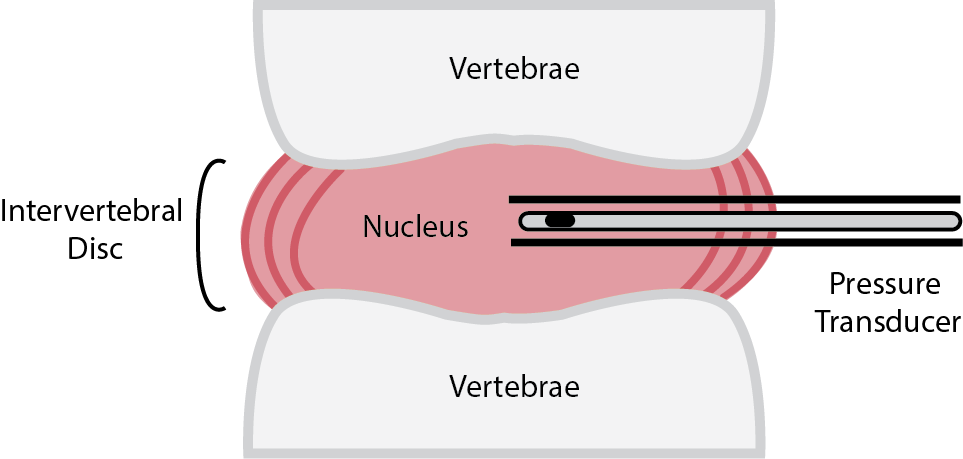
Schematic for measuring intradiscal pressure.
This is possible because Millar Mikro Tip Pressure Catheters can accommodate pressures from -760 mmHg to 4000 mmHg, with a linear response from -50 mmHg to 300 mmHg, since their primary application is the measurement of blood pressure. However, intradiscal pressures are commonly in the range of 1000 - 3000 mmHg, much higher than the usual blood pressure range, therefore pressure measurements must be made in the sensor's non-linear range.
Fortunately, our PowerLab and LabChart data acquisition system options provide support to handle non-linear measurements. Multi-point calibration over the entire pressure range (0 -4000 mmHg) can easily be done within our analysis software LabChart using the Multi-Point Calibration Extension.
It's also important to note that Millar's Pressure Signal Conditioner, which can be used to connect a pressure catheter to an Instrument Interface or PowerLab, has a maximum pressure sensitivity of ~ 500 mmHg. Therefore, to allow for pressure measurements outside of this range (such as intradiscal pressures), a Bridge Amp connected to an Instrument Interface, PowerLab and LabChart should be used.
For more information about measuring intradiscal pressure in animals, check out the publications below.
Publications citing Millar technology for measure intradiscal pressure:
- Effects of chondroitinase ABC on intradiscal pressure in sheep (2001) - SPR 407 catheter
- Effect of cervical spine fusion on adjacent-level intradiscal pressure and segmental motion (2002) - SPR 524 catheter
Millar Research Catheters - Accuracy Matters
Millar Mikro-Tip® pressure catheters provide an ideal solution for measuring highly accurate pressures. Whether measuring arterial and ventricular blood pressures, spinal pressures, tumor pressures, or airway pressures, a high-fidelity pressure sensor from Millar is a necessity for your research.

We provide a range of Millar Catheters from sizes 5 F for larger animals (dog, pig, or sheep) to the world’s smallest 1 F catheter for mice studies.
By combining Millar’s highly sensitive, minimally invasive catheters with the precision of PowerLab data acquisition and LabChart data analysis software, you can build a high quality, flexible system that will give you comprehensive data you can trust.
For more information about our solutions for invasive pressure measurement, please get in touch using one of the forms below!
Request demo Request quote Ask a question
Request a quote
Thank you for your interest in Millar. Please complete and submit the form below and an ADInstruments representative will aim to get in touch within one working day.
If you’d like to speak to someone immediately, office contact information is available from the contact page.
Loading...
Ask a question
Thank you for your interest in Millar . Please complete and submit the form below and an ADInstruments representative will aim to get in touch within one working day.
If you’d like to speak to someone immediately, office contact information is available from the contact page.
Loading...
Request a demo
Thank you for your interest in Millar . Please complete and submit the form below and an ADInstruments representative will aim to get in touch within one working day.
If you’d like to speak to someone immediately, office contact information is available from the contact page.
Loading...
Additional resources:
- Surgical instruction video - Invasive left ventricular pressure measurement via carotid in rat »
- Caring for your Millar catheter - Best Practice Tips »
- Troubleshooting your Millar Catheter - Three common problems and how to fix them »
- How to calibrate your Millar Pressure Catheter »
- Invasive Blood Pressure measurement - Fundamentals and best practices »
1. Heldin, C., Rubin, K., Pietras, K. et al. High interstitial fluid pressure — an obstacle in cancer therapy. Nat Rev Cancer 4, 806–813 (2004) doi:10.1038/nrc1456
2. Kim, H., Yu, A.R., Lee, J.J. et al. Measurement of Tumor Pressure and Strategies of Imaging Tumor Pressure for Radioimmunotherapy. Nucl Med Mol Imaging 53, 235–241 (2019). https://doi.org/10.1007/s13139-019-00598-7
3. Greising, S. M., Sieck, D. C., Sieck, et al. Novel method for transdiaphragmatic pressure measurements in mice. Respiratory physiology & neurobiology, 188(1), 56–59 (2013). https://doi.org/10.1016/j.resp.2013.04.018
4. ATS/ERS Statement on respiratory muscle testing. Am J Respir Crit Care Med. 2002;166:518–624.
5. Takahashi, K., Shima, I., Porter, R.W. Nerve root pressure in lumbar disc herniation. Spine 24, 2003-2006 (1999). doi: 10.1097/00007632-199910010-00007
6. Sasaki, M., Takahashi, T., Miyahara, K., Hirose, T. Effects of chondroitinase ABC on intradiscal pressure in sheep. Spine 26, 463-468 (2001).

