The Langendorff Perfusion System
In 1898, Oskar Langendorff published the foundations of the ex vivo Langendorff perfusion technique which is still commonly used today.
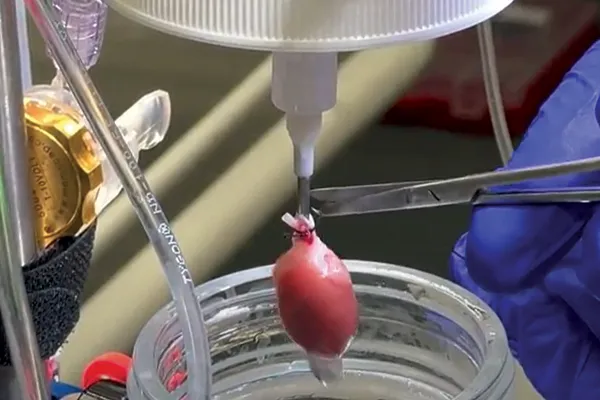
As the perfusate is designed to mimic the qualities of blood - it’s composition is extremely important. Ideally, the perfusate should have a buffering capacity (7.4 pH), energy source, oncotic pressure capacity, oxygen-carrying capability whilst being maintained at physiological temperature (37 C).
Related: 12 pro tips for Langendorff technique >>
So what are some of the perfusate systems you can use?
Dr. Melanie White, cardiovascular researcher and pathology lecturer at the University of Sydney shares her expert knowledge on choosing the correct perfusate system, whether that be crystalloid, whole blood or erythrocyte, when performing a Langendorff isolated heart experiment.
Crystalloid Perfusion
When we talk about crystalloid perfusions, we're mainly referring to Krebs-Henseleit Buffer (KHB). It's the most widely used perfusion buffer associated Langendorff perfusion.

KHB contains ions that mimic the plasma content, such as sodium chloride magnesium sulfate, potassium phosphate, sodium bicarb, and calcium chloride. It’s important that the buffer is bubbled, with not only with oxygen at 95% but also some carbon dioxide, to maintain a physiological pH of 7.4.
It is important to consider the physiological levels of these ions when developing an experimental protocol, as most of the components of the KHB will actually be at supraphysiological levels (higher than that normally found within the body).
The perfusate must also provide an exogenous carbon source for cardiac metabolism - the most popular choice being glucose. Pyruvate and free-fatty acids can also be used as carbon sources, however, you may encounter some issues with solubility and foaming.
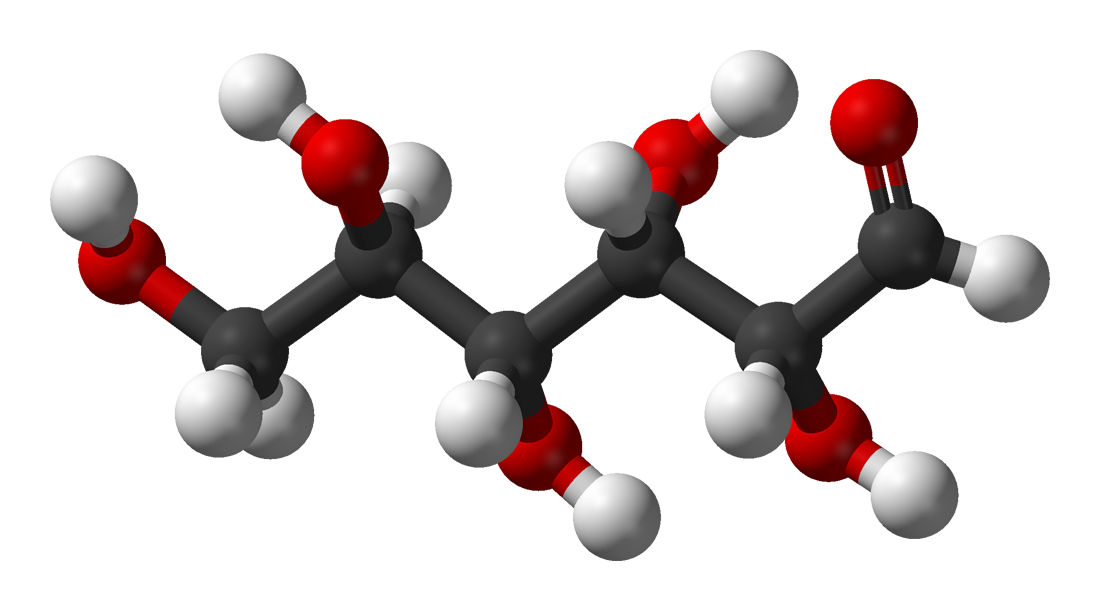
Lastly, given that KHB is a protein-free system, the oncotic pressure is significantly lower than physiological levels, increasing the total tissue water content, effectively simulating an oedemic situation. This is further exacerbated by the supraphysiological flow rates required in the system. In the absence of hemoglobin, the oxygen-carrying capacity to the vascular bed can also be decreased, relatively.
Even in light of these supposed limitations, the crystalloid perfusion system still remains popular, mainly because of the low cost associated and the reproducibility in the production of the buffer systems.
Whole Blood Perfusion
The advantage of a whole blood perfusion system is the ability to maintain an environment that is almost identical to the physiological setting. However, setting it up can be significantly more challenging in comparison to a crystalloid perfusion system.

The main drawback of this system is that it originally suffered from an incapacity to re-oxygenate the blood after ejection from the ex vivo coronary circulation. To circumvent such an issue, a donor animal can be used to re-oxygenate the blood. However, this recirculation also means that the blood must be inline filtered, which can pose additional issues.
Flow rates in the whole blood perfusion system are significantly lower than crystalloid, however, there is a significant risk of hemolysis or blood cell breakdown, and the humoral effects of the donor animal can have an influence on the ex vivo heart.
Conversely, when we consider that the perfusate coming back from the ex vivo heart may contain contaminants, this can have an influence on the donor animal, and these are considerations that need to be addressed as factors when developing experimental protocols.
Erythrocyte Perfusion
Erythrocyte perfusion uses a mix of bovine sources for red blood cells and albumin, in addition to a crystalline buffer such as KHB.
This results in a perfusate system (like whole blood perfusion) that is able to maintain physiological osmolality as well as oncotic pressure.
Hearts that are subjected to erythrocyte perfusion tend to maintain physiological perfusion rates for a significantly longer period of time. However, once again there's an incremental cost increase in this system and it's an incredibly laborious procedure to make the perfusate.
Hopefully, this summary has helped you decide which is the best perfusate for your isolated Langendorff heart system. For more information about the Langendorff perfusion technique, check out Dr. Melanie White's best practices and key considerations when setting-up an isolated Langendorff heart preparation.
Related:
Langendorff vs Working Heart Perfusion – What’s the difference?
An introduction to the isolated Langendorff heart technique (free on-demand webinar)
Effects of constant flow vs. constant pressure perfusion on Langendorff isolated heart studies
Our isolated heart systems - Simplicity and flexibility for cardiovascular and pharmacology research
Three benefits of isolated organ research
Key considerations when performing and isolated perfused liver experiment

